About
At UC Riverside (UCR), the Research Compliance supports the Conflict of Interest (COI) Committee, which advises the Vice Chancellor for Research and Economic Development (VC-RED). Together, they ensure that research conducted at UCR is designed, conducted, and reported objectively—free from bias and aligned with the highest ethical standards.
The COI team acts as a central liaison among faculty, staff, sponsors, and administrative offices to facilitate the disclosure, review, and management of real or perceived conflicts of interest in research.
As part of the University of California system, UCR adheres to system wide policies addressing financial conflicts of interest, including compliance with State of California and Federal regulations. These policies promote research integrity while supporting the University’s mission to transform lives through discovery, communication, translation, application, and preservation of knowledge.
Conflict of Interest
At UCR, a COI in research occurs when an investigator has an outside interest, generally financial, that is related to their research or institutional responsibilities and could directly and significantly affect, or appear to affect, the design, conduct, or reporting of that research.
These outside financial interests include but are not limited to:
- Income from consulting or speaking engagements
- Equity or stock ownership
- Patents, royalties, or other intellectual property rights
- Sponsored or reimbursed travel
- Paid or unpaid positions in external organizations
These interests also include financial holdings of the investigator’s spouse, registered domestic partner, and dependent children.
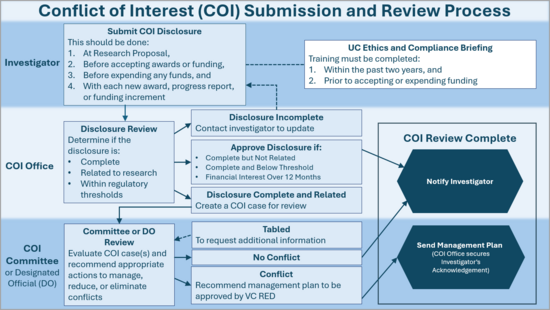
To ensure transparency and uphold the integrity of research, investigators are required to disclose all related financial interests in the Kuali Research COI (KCOI) system. The COI Committee reviews these disclosures to determine whether a related Significant Financial Interest (SFI) could directly and significantly affect the design, conduct, or reporting of funded research.
This process helps UCR uphold ethical research practices and maintain research integrity and public trust.
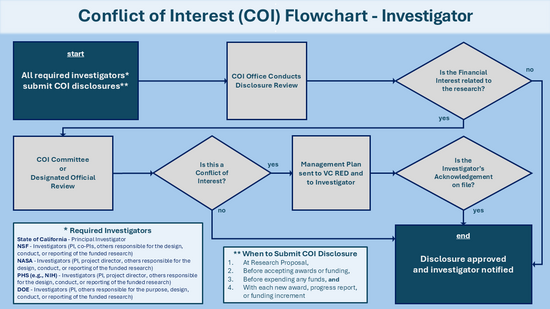
All required investigators must submit their related financial interest disclosures in KCOI. KCOI provides the necessary disclosure forms and ensures compliance with sponsor-specific requirements including those from State, PHS, DOE, NSF, and NASA.
To complete a disclosure submission:
- Log in to the KCOI Dashboard.
- Click the “Update Disclosure” button in the upper right.
- Use right-sidebar navigation (Next, Save, etc.) to complete each section.
- On the final page, click “Submit” in the right sidebar to finish your disclosure.
Research Compliance staff at UCR conduct the initial review of disclosure forms submitted in KCOI. If they determine that an investigator’s significant financial interest is related to their research, the disclosure is forwarded to the COI Committee.
The COI Committee reviews the financial interest alongside the research at its monthly meeting. If the committee determines that the financial interest constitutes a conflict of interest, meaning it could directly and significantly affect the design, conduct, reporting, or purpose of the research, they recommend an appropriate management and monitoring plan. This plan is then submitted to the VCRED for approval.
Researchers are encouraged to submit disclosure forms well in advance of the expected research start date to allow sufficient time for review and approval of potential issues.
Initial Review
- Disclosures are reviewed within 3–5 business days.
- Investigators will be contacted by email if additional information or further review is required.
Committee Review
- If a disclosure requires committee review, the review process typically takes 4–6 weeks.
- The timeline depends on:
- The date the initial disclosure was submitted, and
- How quickly requests for additional information are received.
Committee Meetings
- The committee generally meets on the last Friday of each month.
- Meeting dates may shift when necessary (e.g., holidays or scheduling adjustments).
Policy and Regulatory Requirements
UCR adheres to five regulatory requirements for reviewing and reporting financial conflicts of interest in research, all of which promote transparency, objectivity, and compliance with University of California, State, and Federal regulations and policies. The disclosure chart explains what triggers a disclosure, who is required to disclose a financial interest, when disclosures must be made, where disclosures should be submitted, and what training is necessary to remain in compliance.
Investigators must disclose their financial interests received within the 12 months prior to disclosure submission including the financial interests of their immediate family member. All disclosures must be approved prior to acceptance of the award or additional funding, and/or to expenditure of funds.
-
State Law
Reporting Triggers - Research contract/grant from a non‐governmental entity as the prime and/or direct sponsor.
- Research Gifts earmarked for a specific individual or specific research project.
- Material Transfer Agreement
Exclusions:
- All exempt sponsors: FPPC exempt list
- All US non-profit, tax-exempt educational institutions.
Who Must Disclose UCR Principal Investigator and Co-Principal Investigator, if applicable When Disclosure is Required - With the initial award
- With renewal award (additional funding intended to extend or results in the extension of a project beyond the originally approved project period)
What Must Be Disclosed A financial interest in the non‐governmental sponsor of the research including:
- Income ≥ $500
- Investments ≥ $2,000
- Director, Officer, Employee, Partner, Trustee, Consultant, or Management position
- Loan Balances ≥$500
- Personal Gifts Valued at ≥ $50
- Travel Reimbursements
Form Location Kuali Research COI (KCOI)
For Gifts only: VCUATraining Requirement All extramurally funded researchers are required to complete the UC Ethics and Compliance Briefing for Researchers (ECBR) training, as mandated by UC Office of the President (UCOP). Must be renewed every two years. -
National Science Foundation (NSF) and National Aeronautics and Space Administration (NASA)
Reporting Triggers - NSF research contracts and grants
- NASA research contract and cooperative agreements
- Non‐governmental entity which flows down NSF/NASA funds (including SBIR/STTR subawards)
- California Institute for Regenerative Medicine (CIRM) Grant, Juvenile Diabetes Research Foundation (JDRF) Grant
- UC Special Research Programs (HIV/AIDS, breast cancer and tobacco related disease)
Who Must Disclose UCR Investigator(s) (individuals responsible for the design, conduct, or reporting of the funded research project) When Disclosure is Required - With the initial proposal
- When a new Investigator(s) is added
- With any change in an investigator’s financial interest
What Must Be Disclosed A significant financial interest related to the research project including:
- Salary, honoraria, royalties, venture or other capital financing, or other payments > $10,000 (excluding income from seminars, lectures, teaching engagements, or service on advisory committees or review panels for public or nonprofit entities)
- Equity interest > $10,000 or at least 5% ownership interest
- Intellectual Property interest in a patent, patent application, or copyright of a software (excluding intellectual property owned by the UC.)
Form Location Kuali Research COI (KCOI) Training Requirement All extramurally funded researchers are required to complete the UC Ethics and Compliance Briefing for Researchers (ECBR) training, as mandated by UC Office of the President (UCOP). Must be renewed every two years. -
Public Health Service (PHS) and Department of Energy (DOE)
Reporting Triggers - PHS grant, cooperative agreement, contract
- DOE financial assistance award (e.g., grant, cooperative agreement or technology investment agreement)
- Non-governmental entity which flows down PHS/DOE/USDA funds
- Research Funding from organizations that follow PHS disclosure requirements (i.e., American Cancer Society, American Heart Association)
Exclusions:
- SBIR Phase I or STTR Phase I award
- Office of Indian Energy
Who Must Disclose Investigator(s): Individuals responsible for the design, conduct, purpose*, or reporting of the funded research project.
*For DOE only
When Disclosure is Required - With the initial and renewal proposals
- With supplemental funding proposal
- With non‐competing continuation
- With no cost time extension
- When a new Investigator(s) is added
- At least annually for duration of project
- With any new significant financial interest (New significant financial interests must be reported within 30 days of acquiring or discovering the new SFI.)
What Must Be Disclosed A significant financial interest related to the Investigator’s institutional responsibilities including:
- If publicly traded entity, total value of compensation and equity interest* > $5,000
- If non-publicly traded entity, total value of compensation > $5,000 or any equity interest*
- Income received related to intellectual property > $5,000 (excluding intellectual property owned by the UC)
- Any reimbursed or sponsored travel, excluding:
- Travel that is reimbursed or sponsored by a US Federal, state, local government agency, a domestic Institution of higher education, or a research institute affiliated with an Institution of higher education within US (for PHS only- also exclude an academic teaching hospital, a medical center); and
- Sponsored or reimbursed travel for the Investigator’s spouse/registered domestic partner and/or dependent children.
*Equity interests includes stock, stock options, private equity and other ownership interests
Form Location Kuali Research COI (KCOI) Training Requirement All extramurally funded researchers are required to complete the UC Ethics and Compliance Briefing for Researchers (ECBR) training, as mandated by UC Office of the President (UCOP). This requirement applies to all funding sponsors including PHS and DOE, and must be renewed every two years.
Conflict of Interest (COI) FAQs
-
Conflict of Interest
Q: What are some examples of mitigation or management strategies for COIs?
A: COIs can be managed, reduced, or eliminated through measures such as:- Public disclosure of significant financial interests
- Training in research-related COI for all research personnel
- independent monitoring of research
- Using randomized or double-blind study designs
- Modifying the research plan to ensure objectivity
- Stepping down as principal investigator
- Restricting involvement in parts of the research
- Divesting the financial interest
- Ending relationships that create the conflict
Q: My disclosure will be sent to COI for review. When will I receive the results so my study funds can be released?
A: Reviews typically take four to six weeks, depending on the nature of the disclosure. ORC staff will contact you if additional review is needed. -
Conflict of Commitment
Q: How are Research-Related Conflicts of Interest (COI) and Conflicts of Commitment (COC) related?
A: Both COI and COC may involve an Investigator’s outside financial interests and institutional responsibilities, but they are managed separately on campus under different offices, policies, and procedures.- Research-Related COI:
Occurs when an investigator’s outside financial interests or obligations—such as salaries, consulting fees, equity ownership, intellectual property income, gifts, loans, travel payments, or roles like director or consultant—could bias or appear to bias a research project. This includes interests held by the investigator’s immediate family. Contact the COI office for more information. - Conflict of Commitment:
The UC Regents require that faculty not allow outside activities to interfere with primary University duties. Relevant policies are APM 025 (general campus faculty) and APM 671 (Health Sciences Compensation Plan), both of which set limits on outside professional activities.
To comply, UCR faculty must:- Obtain Prior Approval for applicable outside activities and
- Submit an Annual Report
For details, see the Office of Academic Personnel’s COC page.
- Research-Related COI:
-
Founding or Advising a Company
Q: What general principles should I keep in mind when starting a company?
A:- Keep your company activities separate from UC research and administrative duties.
- Your company will be treated the same as any external company.
- Always disclose potential conflicts so COI can assess and mitigate risk.
- Many research-related conflicts are manageable, not prohibited.
- Human-subjects research receives heightened COI review.
- Conflicts must be evaluated on a case by case basis.
- When unsure, disclose it to COI.
- Using a non-conflicted investigator and study blinding may help, but COI must still review the situation.
Q: Can I start a company?
A: Yes. UCR’s conflict of interest (COI) and conflict of commitment (COC) policies do not prohibit investigators from forming companies. However, disclosures and approvals may be required. For entrepreneurial resources, visit UCR’s EPIC (Entrepreneurial Proof of Concept and Innovation Center).Q: Can I serve on a board of directors?
A: Serving on a Board of Directors, Advisory Board, or similar entity is not prohibited.- If the role includes financial interests (e.g., equity, compensation), COI disclosure may be required.
- If the COI Committee determines a conflict exists in relation to your research, management steps may be required.
- Board service carries fiduciary—not operational—responsibility, so it is permissible.
- Your primary professional commitment must remain with the University. More on COC is available on the Conflict of Commitment and Outside Professional Activities page.
Q: Can I serve on a scientific advisory board?
A: Serving in this capacity in permitted, as these roles do not imply management responsibility.- You must recuse yourself from decisions involving funding of your own research.
- If financial interests are involved, COI disclosure may be required.
Q: I consult for a company that wants to fund my UCR lab through an industry-sponsored research agreement. Is this allowed?
A: Strategies and review factors include:- Use a non conflicted investigator and/or blind the study.
- COI will assess issues such as:
- Who supervises research personnel.
- Whether any personnel report to the conflicted researcher.
- Whether a non conflicted investigator reports to the conflicted individual.
- Whether students are involved and who supervises them.
- Who oversees data collection, analysis, and reporting.
- Whether human subjects are involved.
- Additional measures may be required depending on the level of risk and study design.
Q: I am a co-founder of a company that wants to fund my UCR lab through an industry-sponsored research agreement. Is this allowed? What should I be concerned about?
A: Some important safeguards to consider:- You cannot represent both the University and the company during contract negotiations.
- Your company should use an external attorney or a representative with no UCR affiliation.
- If you have a financial interest and serve as PI or Co PI, you must disclose it for COI review.
Q: I consult for a company that wants to fund my UCR lab through an industry-sponsored research agreement. Is this allowed?
A: Companies may fund your lab.- If you have a financial interest in the sponsor and are PI or Co PI, the financial interest will require COI disclosure and review before the research can proceed.
-
Intellectual Property and Technology
Q: Can I participate in research sponsored by a company that licensed technology I invented at UCR?
A: Yes, but it may create a research-related conflict of interest if you have a financial interest in the sponsoring company. If you do, you may need to disclose it.
For specific disclosure thresholds, see the State Law portion of COI's Policy and Regulatory Requirement section.Q: I am an inventor on a university-owned patent. Can I serve as an investigator on research testing its effectiveness?<
A: Possibly. It depends on:- Who is sponsoring the research, and
- Whether you have a financial interest in the sponsor or the research.
If you do have a financial interest, the situation may require review by the COI office.
-
Equity
Q: May I own stock in a publicly traded company and still serve as an investigator on a study of that company’s products?
A: Yes, but depending on the sponsor and the amount or value of your stock, you may be required to disclose this interest and undergo a COI review.
Refer to the State Law portion of COI's Policy and Regulatory Requirement section for threshold details.
Q: I have equity in a company that wants to fund my UCR lab through an industry-sponsored research agreement. Is this allowed?
A: You may hold equity in the company funding your lab. However, if you are an investigator and your financial interest meets sponsor disclosure thresholds, COI review and approval may be required.
Refer to the State Law portion of COI's Policy and Regulatory Requirement section for threshold details.
Office Hours
- When: First Thursday of each month (dates are subject to change), 9:30 AM - 10:30 AM
- Meeting ID: 912 9637 1281
- Passcode: 868913
If you need to meet outside these scheduled office hours, please contact us to arrange an appointment.
Contact Us
For any COI related questions, please contact Research Compliance as soon as possible. Email coi@ucr.edu to set up an appointment.